General Information:
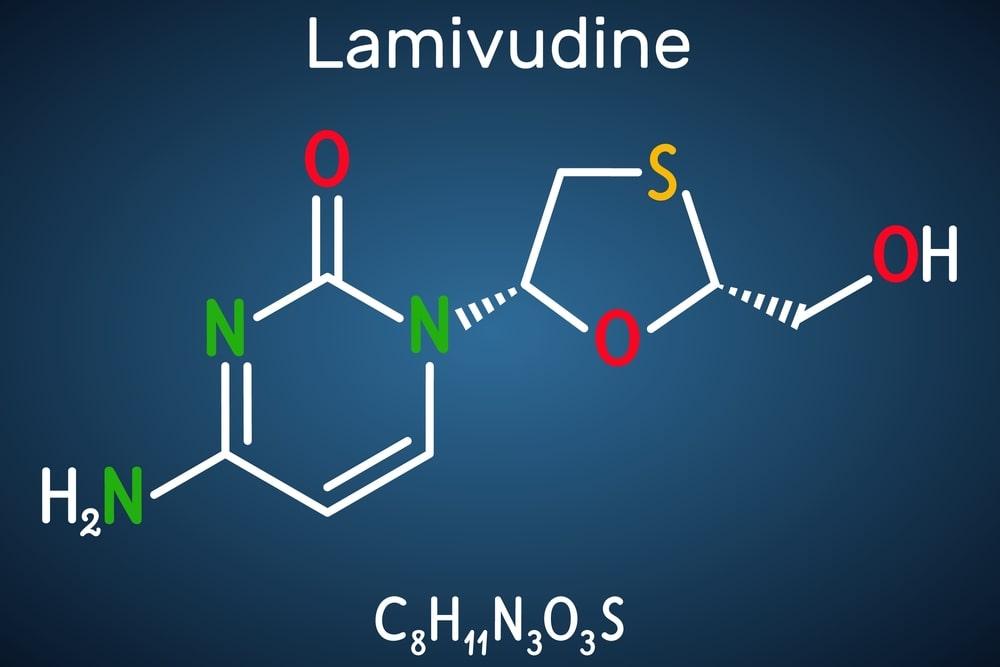
Lamivudine, also termed 2′,3′-dideoxy-3′-thiacytidine, or 3TC. It is in the nucleoside reverse transcriptase inhibitor (NRTI) group of medications and is widely used in the management and treatment of HIV-1 and hepatitis B. It is a valuable agent in the treatment of HIV (and other disorders when applicable). This drug is available in 150 mg scored oral tablets, 300 mg tablets, and 5 mg or 10 mg/ml oral solutions. It may be taken with or without food/meal and needs to be stored at room temperature. Lamivudine 300 mg/150 mg should not be administered to those with known hypersensitivity. Headache, nausea, vomiting, fever, diarrhea, abdominal pain, weight loss, cough, and nasal signs and symptoms are the commonly reported side effects of the medicine.
Mechanism of Action:
Lamivudine is formulated as a dideoxynucleoside cytosine analog that blocks/inhibits viral DNA synthesis via reverse transcriptase DNA chain termination post phosphorylation. Once inside the cell, lamivudine, 2′-deoxy-3′-thiacytidine, is metabolized to the triphosphate form, lamivudine triphosphate (3TC-TP or L-TP), and the monophosphate form, lamivudine monophosphate (3TC-MP or L-MP), during intracellular kinase phosphorylation. Both L-TP and L-MP forms inhibit viral DNA synthesis.
It is beneficial as an antiviral medication because it is primarily not identified by human polymerase as a substrate. As an L-nucleoside enantiomer instead of a D-enantiomer, active lamivudine is not primarily identified by human polymerases as a substrate but actively competes with natural cytidine triphosphate to block reverse transcriptase DNA synthesis noticed in both HBV and HIV-1 infection. The triphosphate metabolite can also weakly block mammalian DNA polymerase (α and β types) and mitochondrial DNA (mtDNA) polymerase.
Lamivudine is absorbed quickly, with a highest lamivudine concentration of 0.5-1.5 hours following oral dose administration and absolute bioavailability of roughly 82% in adults and 68% in children. It can be used with or without a meal. While systemic exposure to this medicinal product does not change with food intake, taking it with food slows the absorption rate by 40 percent. Manufactured as an oral liquid and tablet form, Lamivudine has equitable bioavailability in adult patients.
This medication is distributed into the total body fluid or extravascular spaces. It has a long intracellular half-life of as long as 15.5 hours for HIV-positive cells and 19 hours for HBV-positive cells. However, viral replication site reservoirs demonstrated varying pharmacokinetics. This therapeutic drug freely traverses the placenta, often existing with exceedingly elevated concentrations, and readily exists in breast milk.
References: